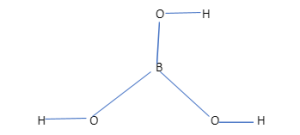
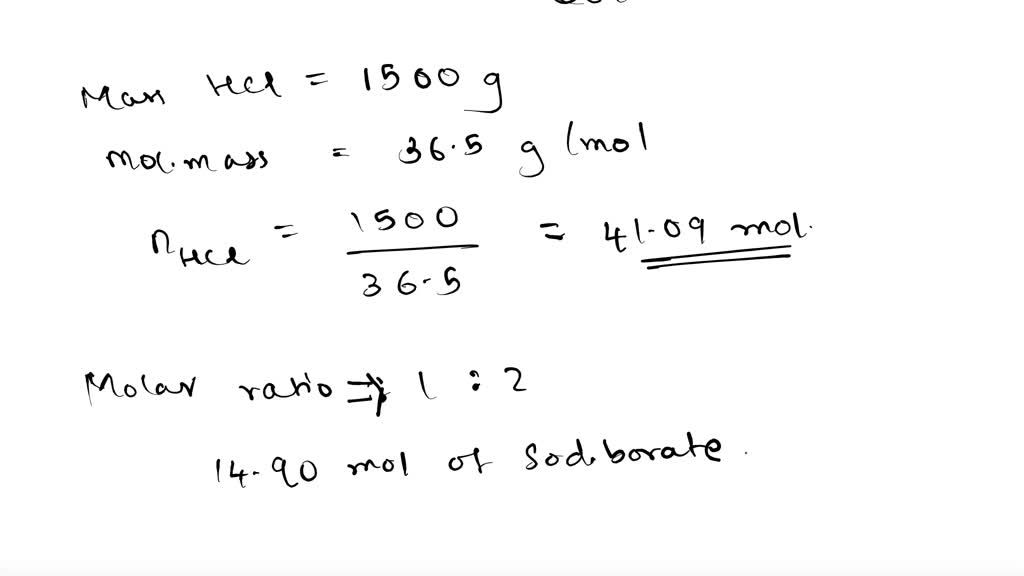SOLVED: Boron (B), one of the constituent elements of boric acid, naturally occurs as isotopes of 10B and 11B with percent natural abundance as 20% and 80%, respectively. Boric acid (H3BO3) is

Inorganic Group Lab report - Preparation and Analysis of Boric acid and Sodium Perborate NAME: Brian Chung Young Min Hong Date submitted: | Course Hero

Ultralong Room‐Temperature Phosphorescence from Boric Acid - Zheng - 2021 - Angewandte Chemie International Edition - Wiley Online Library

PDF) Novel (Glycerol)borate-Based Ionic Liquids: An Experimental and Theoretical Study | Fabio Bellina - Academia.edu
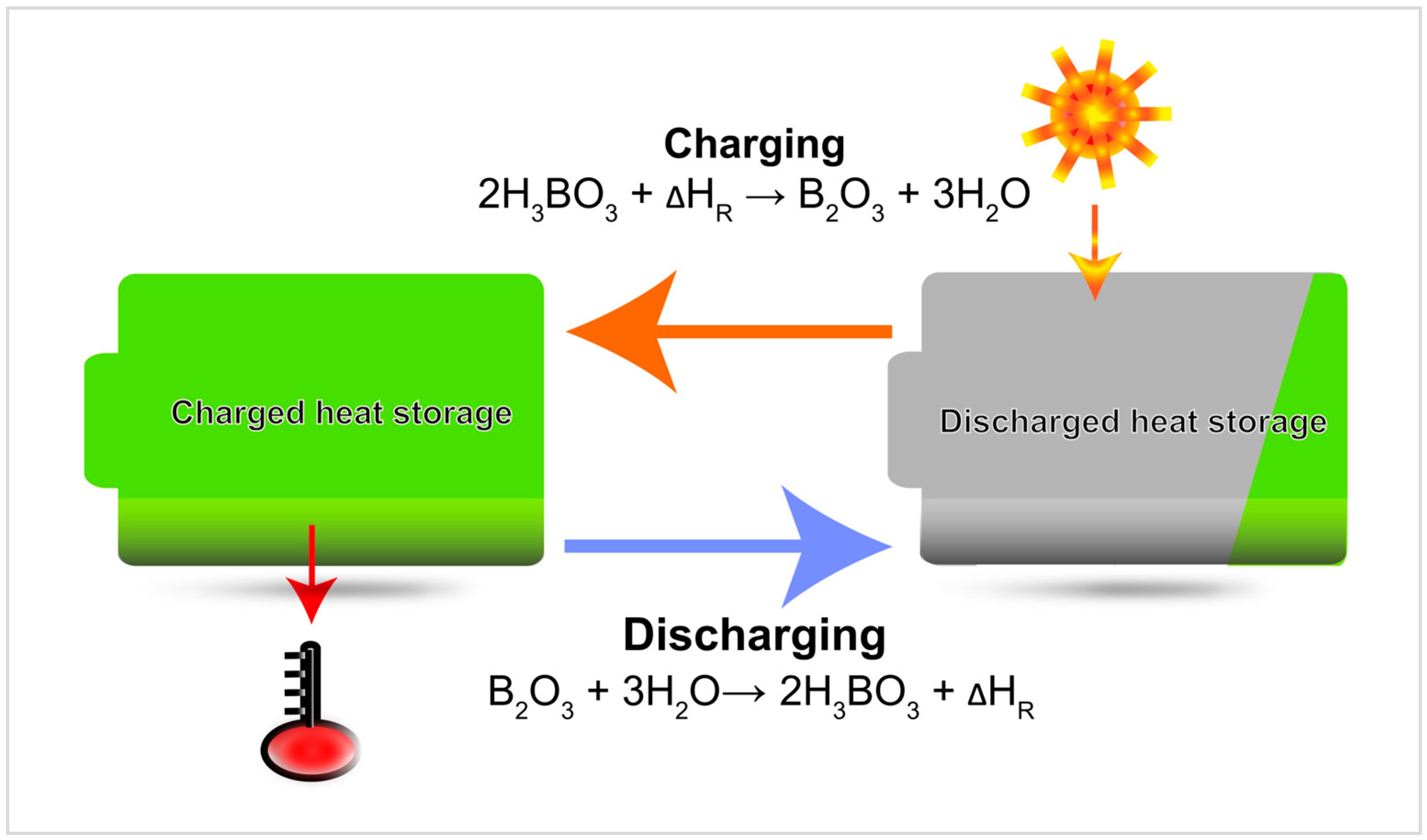
Energies | Free Full-Text | Boric Acid: A High Potential Candidate for Thermochemical Energy Storage

A solution of 8% boric acid is to be diluted by adding a 2% boric acid solution to it. The resulting mixture is to be more than 4% but less than 6%

Optimization of boric acid extraction from ulexite mineral by using supercritical carbon dioxide - ScienceDirect